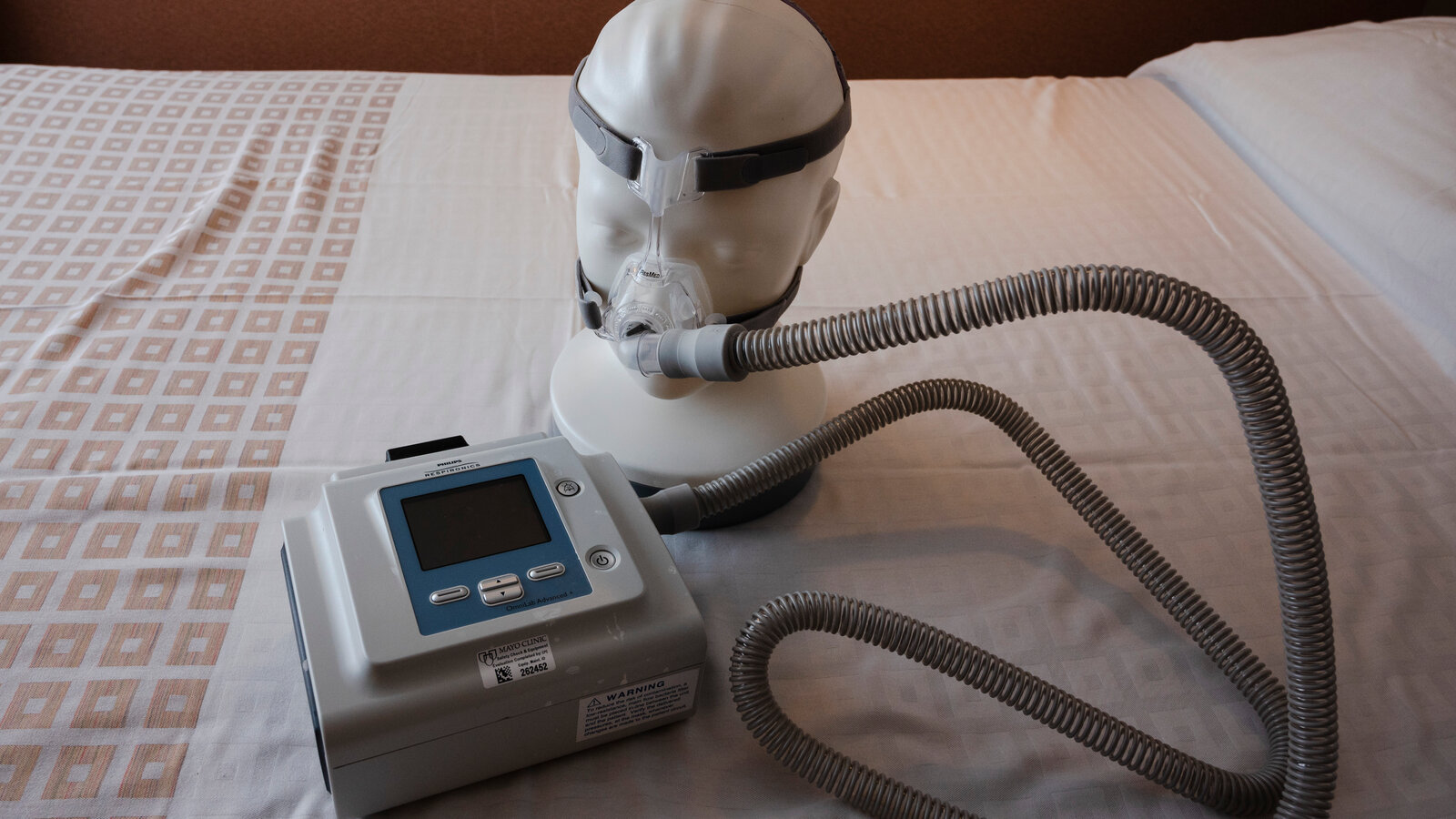
When a device designed to help people breathe at night becomes a safety story, it forces a hard look at how medical products are made, monitored, and recalled. The Philips CPAP Recall, affecting millions of CPAP, BiPAP, and ventilator units, put patient safety and corporate responsibility under a spotlight and raised a practical question: what rights do patients have when a device meant to help instead puts them at risk? Philips CPAP recall developments continue into 2025, and the legal landscape around medical device recalls remains active and evolving.
Medical device recalls — such as the Philips CPAP case — raise serious concerns about patient safety and corporate responsibility. This article explains the legal implications of defective products and outlines the rights of affected patients. Readers gain insight into how such recalls fit within the broader framework of product liability law.
Overview of the Philips CPAP recall and its health risks
Philips Respironics announced a large-scale recall in June 2021 after identifying that the polyester-based polyurethane (PE-PUR) foam used for sound abatement in certain CPAP, BiPAP, and ventilator models could degrade. Heat, humidity, and especially the use of ozone-based cleaners were linked to breakdown of the foam, potentially releasing black particulates and volatile organic compounds (VOCs) into the airflow pathway. The U.S. Food and Drug Administration (FDA) classified the action as a Class I recall, the most serious, because exposure could cause harm.
Reported and potential health effects include airway irritation, coughing, chest tightness, headaches, dizziness, nausea, sinus and respiratory infections, and potential toxic and carcinogenic effects from long-term exposure to certain chemicals. Some users reported seeing dark particles in the tubing or mask. While not everyone experienced symptoms, the risk profile was significant enough that therapy decisions needed to be individualized with a clinician.
Philips developed a remediation program to repair or replace affected devices and issued updated guidance about device cleaning and continued use, emphasizing consultation with a treating physician, especially for patients with severe sleep apnea who face risks if therapy is abruptly stopped. As of 2024, Philips Respironics agreed to a consent decree with the U.S. Department of Justice to address quality system issues and oversight: regulatory scrutiny remains high as remediation and litigation proceed into 2025. Throughout, the Philips CPAP recall has served as a high-visibility case study in how safety issues can cascade across a sprawling device population.
Patient rights when defective devices cause harm
Patients have clear rights when a defective medical device causes injury or raises credible safety concerns. Those rights flow from a mix of FDA rules, state product liability law, and basic consumer protections.
Key rights include:
- The right to timely, accurate notice of recalls and risks from manufacturers, durable medical equipment (DME) suppliers, and healthcare providers.
- The right to a medical evaluation to assess symptoms possibly connected to foam particulates or VOC exposure, and to discuss whether to continue, modify, or pause therapy.
- The right to repair, replacement, or refund when offered in a recall remediation program, subject to program criteria.
- The right to report adverse events to FDA MedWatch, which helps regulators track patterns and enforce corrective action.
- The right to preserve evidence, the device, tubing, filters, serial number, and all paperwork, if harm is suspected.
- The right to pursue legal remedies for injuries, including claims alleging design defect, manufacturing defect, and failure to warn.
Practically speaking, patients strengthen their position by documenting symptoms, keeping device serial numbers and registration confirmations, and saving receipts for replacement machines, supplies, and medical visits tied to recall-related concerns.
Corporate accountability in medical device recalls
Medical device manufacturers carry legal duties that start long before a recall and continue long after. Under FDA’s Quality System Regulation (21 CFR Part 820), companies must design and validate safe products, maintain robust complaint handling and corrective-and-preventive-action (CAPA) systems, and continuously monitor post-market performance. They must also report adverse events under 21 CFR Part 803 and initiate and manage recalls in line with 21 CFR Part 7.
In the Philips CPAP recall, regulators scrutinized how risk signals were assessed and communicated, the speed and scope of corrective actions, and the effectiveness of the repair-and-replace program. The 2024 consent decree imposed third-party oversight and restrictions until quality and compliance benchmarks are met, an example of how enforcement can reshape corporate processes.
Accountability also means transparency. Clear recall notices, realistic timelines, and accessible reimbursement pathways help patients make informed decisions. Where warnings arrive late or speak too softly to known risks, companies open themselves to claims for failure to warn and punitive exposure when conduct suggests reckless indifference. The broader lesson: in life-sustaining or health-critical products, conservative safety margins and candid communication are not just good practice, they’re legal necessities.
Legal remedies under product liability law in New York
New York law provides multiple pathways for patients who suffered harm linked to recalled medical devices.
Core theories of recovery:
- Design defect (strict liability): Plaintiffs argue a product was unreasonably dangerous as designed and that a feasible, safer alternative existed. New York applies a risk–utility balancing test.
- Manufacturing defect: The specific unit deviated from its intended design or specifications, causing harm.
- Failure to warn: Inadequate warnings or instructions about known or reasonably knowable risks at the time of sale or during post-market life.
- Negligence: Breach of a duty of reasonable care in design, testing, quality control, or post-market surveillance.
- Breach of warranty: Claims may include implied warranty of merchantability and fitness for a particular purpose.
Limitations and timing:
- Personal injury claims are generally subject to a three-year statute of limitations (CPLR 214). For toxic exposure and latent harm, the period can run from discovery of the injury (CPLR 214-c). Wrongful death claims typically must be filed within two years (EPTL 5-4.1).
- New York follows pure comparative fault (CPLR 1411), which can reduce but not bar recovery based on a plaintiff’s share of responsibility.
- Standalone medical monitoring without present physical injury is not recognized under New York law (Caronia v. Philip Morris USA, Inc.).
Procedurally, many Philips CPAP cases have been centralized in a federal multidistrict litigation (MDL No. 3014) in the Western District of Pennsylvania, while individual claims still apply New York substantive law when appropriate. New York-focused firms, including the Jacob Fuchsberg Law Firm, have been active in advising residents on eligibility, evidence preservation, and filing choices, coordinating with the MDL where required.
Compensation avenues for affected patients in 2025
As of 2025, compensation for those affected by the Philips CPAP recall can flow through several channels, depending on the type of harm and documentation available.
Potential avenues include:
- Personal injury claims: Damages may cover medical expenses (diagnostics, specialist visits, medications), lost wages or diminished earning capacity, pain and suffering, and, where warranted, loss of consortium and punitive damages.
- Wrongful death claims: Brought by eligible family members or estate representatives under New York law.
- Economic loss reimbursement: Separate from injury, some users pursued recovery for out-of-pocket costs like replacement devices and supplies. In 2023, an economic loss class settlement of approximately $479 million received approval, addressing certain costs for eligible purchasers.
- Proposed personal injury resolution: In 2024, Philips announced a proposed $1.1 billion settlement framework for U.S. personal injury claims, subject to court approval and individual eligibility determinations: it did not constitute an admission of liability. Status updates continue into 2025.
- Recall remediation programs: Manufacturer-run programs may offer repair, replacement, or refunds. Patients should keep serial numbers, registration confirmations, and correspondence to support claims.
Strong documentation remains the throughline: contemporaneous medical records, symptom timelines, device identifiers, purchase proofs, and communications with physicians and DME suppliers. Experienced counsel, such as New York-based teams at the Jacob Fuchsberg Law Firm, can help align evidence with the correct legal pathway, evaluate MDL participation, and ensure deadlines are met.